Question 2:
Figure 2 shows an apparatus set-up to study the electrolysis of aqueous sodium nitrate solution, NaNO3, using carbon electrodes labelled P and Q.
Answer:
(a)(i)
Sodium ion, Na+, hydrogen ion, H+
Figure 2 shows an apparatus set-up to study the electrolysis of aqueous sodium nitrate solution, NaNO3, using carbon electrodes labelled P and Q.
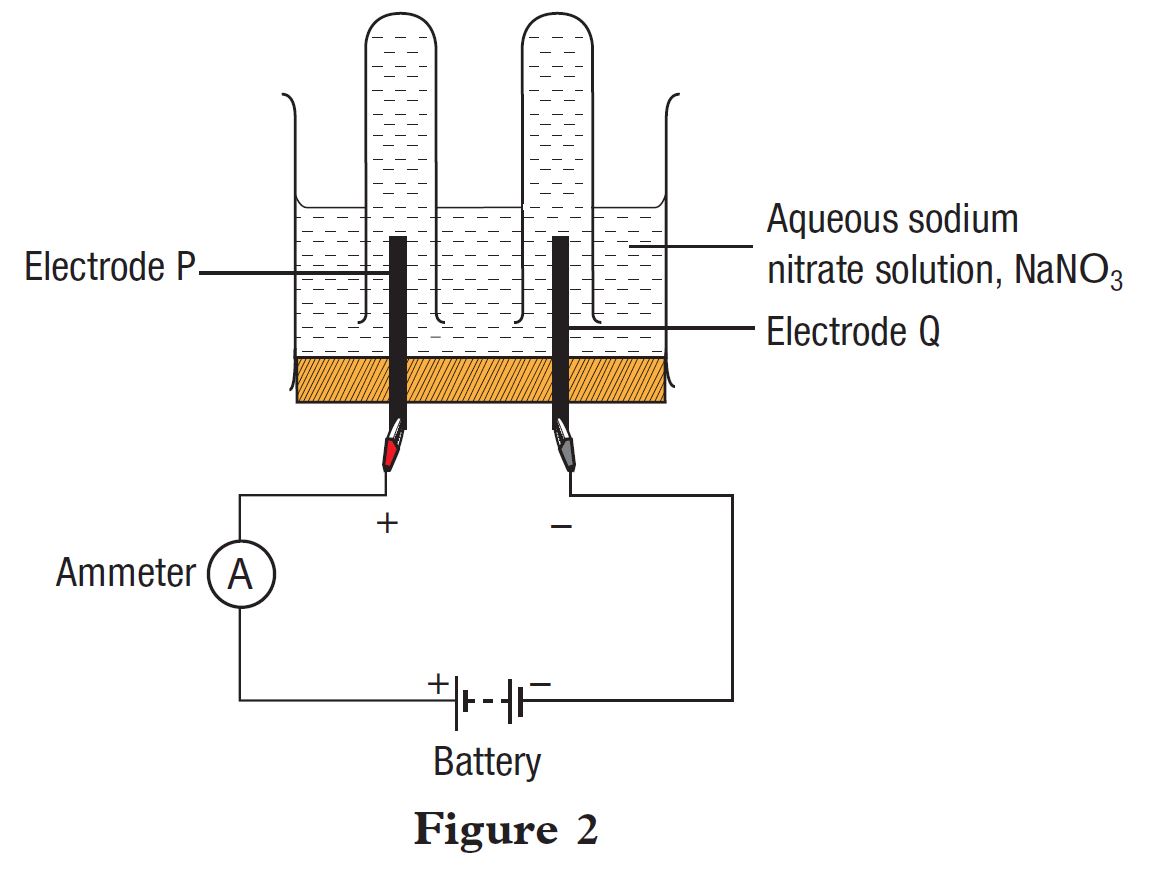
(a)(i) State all the cations present in the electrolyte.
(a)(ii) State all the anions present in the electrolyte.
(b) Which electrode acts as the anode?
(c) Name the ion chosen to be discharged at:
(i) electrode P:
(ii) electrode Q:
(d) Explain your answer in 2(c)(ii) based on the selection of ion to be discharged.
Answer:
(a)(i)
Sodium ion, Na+, hydrogen ion, H+
(a)(ii)
Nitrate ion, NO3–, hydroxide ion, OH–
(b)
Electrode P
(c)(i)
Hydroxide ion
(c)(ii)
Hydrogen ion
(d)
Sodium ions and hydrogen ions move to the cathode. The hydrogen ion is selected to be discharged because it is less electropositive compared to the sodium ion.